How Does Rezūm
Water Vapor Therapy Work?
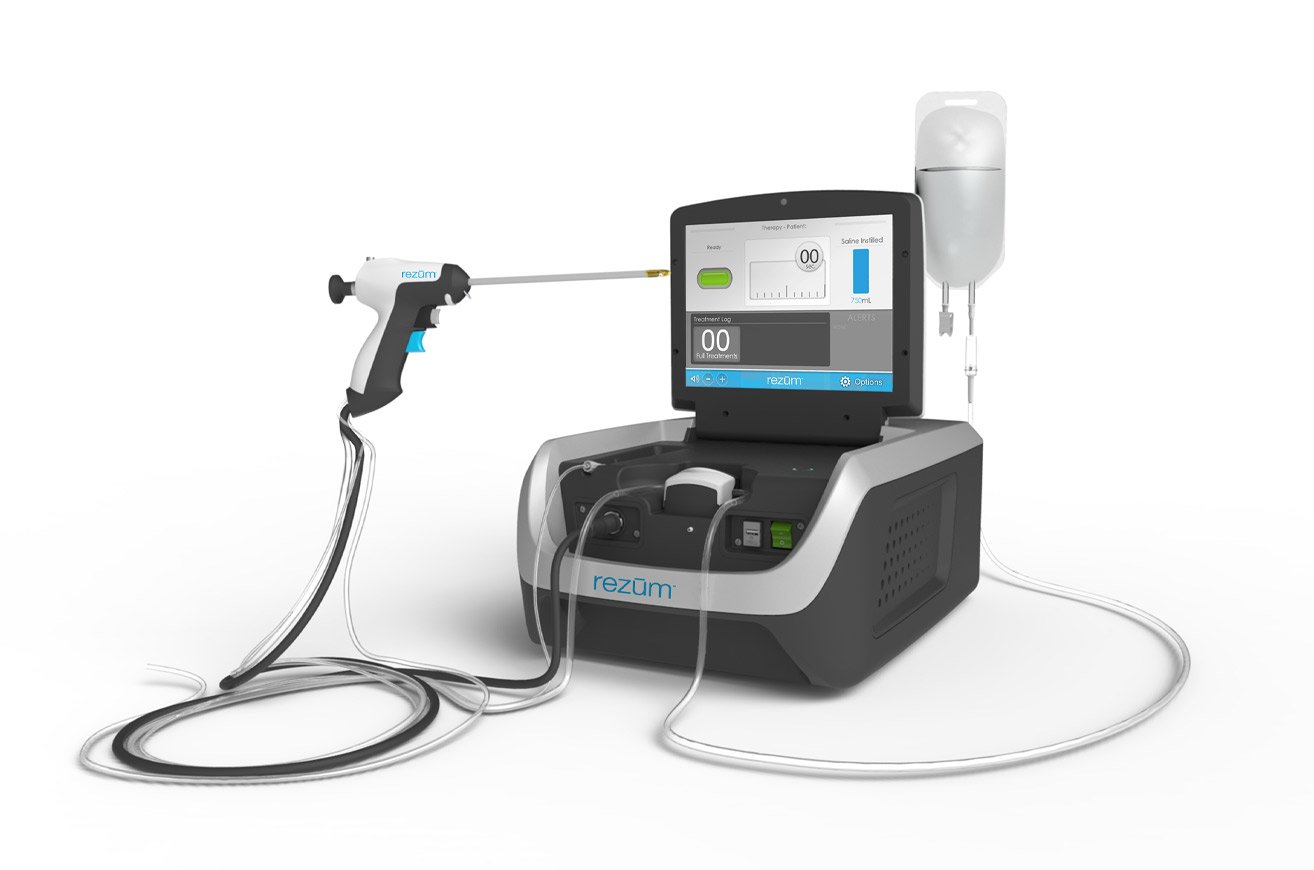
Rezūm harnesses one of nature's most powerful thermodynamic phenomena: the latent heat released by water vapor condensing into liquid inside prostate tissue.
In just 10 minutes of outpatient treatment, water vapor at 103°C is delivered into the obstructive tissue via a transurethral cystoscope. Cell death occurs within the prostate, and the body naturally reabsorbs the destroyed tissue over the following weeks.